You have no items in your shopping cart
Phosphorus pentoxide
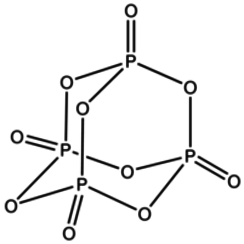
Phosphorus pentoxide is a chemical compound with molecular formula P4O10 (with its common name derived from its empirical formula, P2O5). This white crystalline solid is the anhydride of phosphoric acid. It is a powerful desiccant and dehydrating agent.
Phosphorus pentoxide is a potent dehydrating agent as indicated by the exothermic nature of its hydrolysis:
- P4O10 + 6 H2O → 4 H3PO4 (–177 kJ)
However, its utility for drying is limited somewhat by its tendency to form a protective viscous coating that inhibits further dehydration by unspent material. A granular form of P4O10 is used in desiccators.
Consistent with its strong desiccating power, P4O10 is used in organic synthesis for dehydration. The most important application is for the conversion of primary amides into nitriles:
- P4O10 + RC(O)NH2 → P4O9(OH)2 + RCN
The indicated coproduct P4O9(OH)2 is an idealized formula for undefined products resulting from the hydration of P4O10.
Alternatively, when combined with a carboxylic acid, the result is the corresponding anhydride:
- P4O10 + RCO2H → P4O9(OH)2 + [RC(O)]2O
The "Onodera reagent", a solution of P4O10 in DMSO, is employed for the oxidation of alcohols. This reaction is reminiscent of the Swern oxidation.
The desiccating power of P4O10 is strong enough to convert many mineral acids to their anhydrides. Examples: HNO3 is converted to N2O5; H2SO4 is converted to SO3; HClO4 is converted to Cl2O7; CF3SO3H is converted to (CF3)2S2O5.