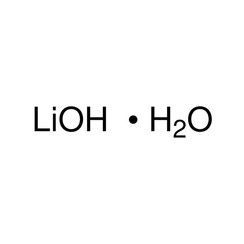You have no items in your shopping cart
Lithium hydroxide
Lithium hydroxide is an inorganic compound with the formula LiOH. It is a white hygroscopic crystalline material. It is soluble in water and slightly soluble in ethanol, and is available commercially in anhydrous form and as the monohydrate (LiOH.H2O). While lithium hydroxide is a strong base, it is the weakest known alkali metal hydroxide.
Lithium hydroxide is mainly consumed in the production of lithium greases. A popular lithium grease thickener is Lithium 12-hydroxystearate, which produces a general-purpose lubricating grease due to its high resistance to water and usefulness at a range of temperatures.
Carbon dioxide scrubbing
Lithium hydroxide is used in breathing gas purification systems for spacecraft, submarines, and rebreathers to remove carbon dioxide from exhaled gas by producing lithium carbonate and water:
- 2 LiOH•H2O + CO2 → Li2CO3 + 3 H2O
or
- 2 LiOH + CO2 → Li2CO3 + H2O
The latter, anhydrous hydroxide, is preferred for its lower mass and lesser water production for respirator systems in spacecraft. One gram of anhydrous lithium hydroxide can remove 450 cm3 of carbon dioxide gas. The monohydrate loses its water at 100–110 °C.
Other uses
It is used as a heat transfer medium and as a storage-battery electrolyte. It is also used in ceramics and some Portland cement formulations. Lithium hydroxide (isotopically enriched in lithium-7) is used to alkalize the reactor coolant in pressurized water reactors for corrosion control.