You have no items in your shopping cart
Potassium hydroxide
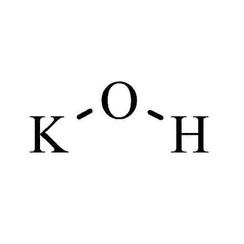
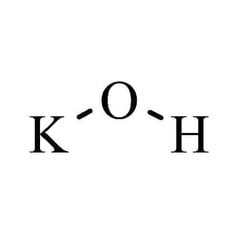
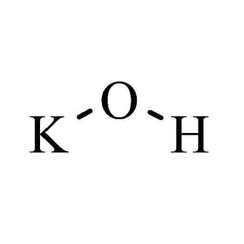
Potassium hydroxide is an inorganic compound with the formula KOH, and is commonly called caustic potash.
Along with sodium hydroxide (NaOH), this colorless solid is a prototypical strong base. It has many industrial and niche applications, most of which exploit its caustic nature and its reactivity toward acids. An estimated 700,000 to 800,000 tonnes were produced in 2005. KOH is noteworthy as the precursor to most soft and liquid soaps, as well as numerous potassium-containing chemicals. It is a white solid that is dangerously corrosive. Most commercial samples are ca. 90% pure, the remainder being water and carbonates.
Uses
KOH and NaOH can be used interchangeably for a number of applications, although in industry, NaOH is preferred because of its lower cost.
Precursor to other potassium compounds
Many potassium salts are prepared by neutralization reactions involving KOH. The potassium salts of carbonate, cyanide, permanganate, phosphate, and various silicates are prepared by treating either the oxides or the acids with KOH.
Manufacture of soft soaps
The saponification of fats with KOH is used to prepare the corresponding "potassium soaps", which are softer than the more common sodium hydroxide-derived soaps. Because of their softness and greater solubility, potassium soaps require less water to liquefy, and can thus contain more cleaning agent than liquefied sodium soaps
As an electrolyte
Aqueous potassium hydroxide is employed as the electrolyte in alkaline batteries based on nickel-cadmium, nickel-hydrogen, and manganese dioxide-zinc. Potassium hydroxide is preferred over sodium hydroxide because its solutions are more conductive. The nickel–metal hydride batteries in the Toyota Prius use a mixture of potassium hydroxide and sodium hydroxide.[18] Nickel–iron batteries also use potassium hydroxide electrolyte.
Food industry
In food products, potassium hydroxide acts as a food thickener, pH control agent and food stabilizer. The FDA considers it (as a direct human food ingredient) as generally safe when combined with "good" manufacturing practice conditions of use. It is known in the E number system as E525.
Niche applications
Like sodium hydroxide, potassium hydroxide attracts numerous specialized applications, virtually all of which rely on its properties as a strong chemical base with its consequent ability to degrade many materials. For example, in a process commonly referred to as "chemical cremation" or "resomation", potassium hydroxide hastens the decomposition of soft tissues, both animal and human, to leave behind only the bones and other hard tissues. Entomologists wishing to study the fine structure of insect anatomy may use a 10% aqueous solution of KOH to apply this process.
In chemical synthesis, the choice between the use of KOH and the use of NaOH is guided by the solubility or keeping quality of the resulting salt.
The corrosive properties of potassium hydroxide make it a useful ingredient in agents and preparations that clean and disinfect surfaces and materials that can themselves resist corrosion by KOH.
KOH is also used for semiconductor chip fabrication. See also: anisotropic wet etching.
Potassium hydroxide is often the main active ingredient in chemical "cuticle removers" used in manicure treatments.
Because aggressive bases like KOH damage the cuticle of the hair shaft, potassium hydroxide is used to chemically assist the removal of hair from animal hides. The hides are soaked for several hours in a solution of KOH and water to prepare them for the unhairing stage of the tanning process. This same effect is also used to weaken human hair in preparation for shaving. Preshave products and some shave creams contain potassium hydroxide to force open the hair cuticle and to act as a hygroscopic agent to attract and force water into the hair shaft, causing further damage to the hair. In this weakened state, the hair is more easily cut by a razor blade.
Potassium hydroxide is used to identify some species of fungi. A 3–5% aqueous solution of KOH is applied to the flesh of a mushroom and the researcher notes whether or not the color of the flesh changes. Certain species of gilled mushrooms, boletes, polypores, and lichens are identifiable based on this color-change reaction.